Heartburn, heavy digestion or abdominal discomfort after eating are symptoms that many people naturally report, but which are rarely investigated in depth. However, behind these seemingly common digestive complaints may lie one of the most widespread chronic bacterial infections on the planet.
Helicobacter pylori bacteria live in the stomach and have the ability to survive in an extremely acidic environment, which is unusual for microorganisms.
According to the neurogastroenterologist Malena García Arredondo, Director of the Digestive Tract and Microbiota Unit MGA Healthy Digest at Publio Cordón Memorial Hospital in Madrid, Many people live with this bacterium for years without knowing it.

The specialist points out that symptoms such as heartburn, a feeling of heaviness or slow digestion are often normalised, when in fact they could be related to alterations in the digestive system. But the story does not end in the stomach.
In recent years, medical research has begun to look at the digestive system in a new way. Not just as an organ responsible for processing food, but as a complex ecosystem made up of trillions of micro-organisms.
This ecosystem - known as the gut microbiome - influences:
- metabolism
- hormone regulation
- inflammation of the body
- communication with the brain.
So even when an infection such as Helicobacter pylori is treated and goes away, some people continue to have digestive symptoms. In many cases, this is linked to the so-called gut-brain axis and changes in the microbiota. And it is this very system that is now appearing in one of the most innovative areas of medicine.
A recent analysis published in the British Journal of Clinical Pharmacology raises a hypothesis that is gaining traction in biomedical research: the response to GLP-1 drugs may depend largely on the gut microbiome.
These drugs - including Semaglutide and Liraglutide - were initially developed to treat type 2 diabetes, but are now changing the approach to obesity and cardiovascular risk.
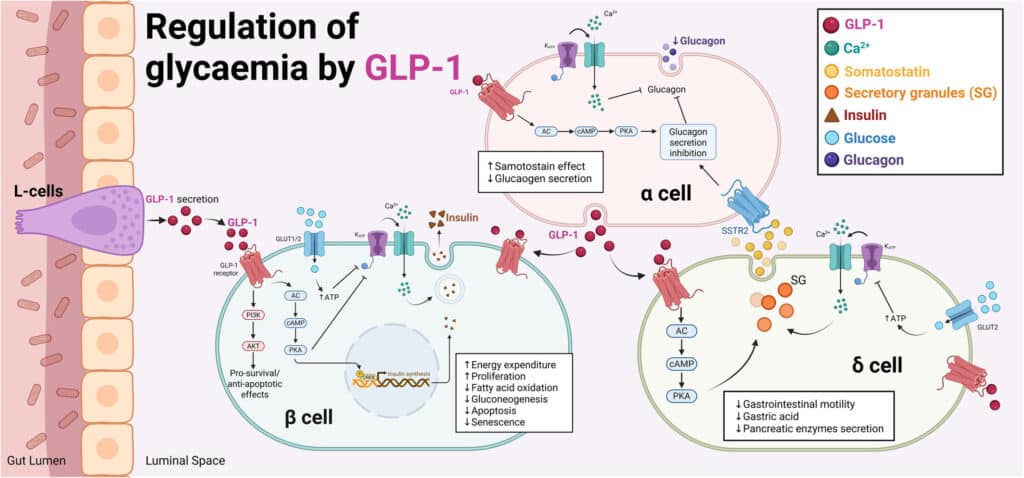
They work by mimicking a hormone that the body produces itself to produce:
- regulating blood sugar
- slowing down digestion
- sending satiety signals to the brain
However, researchers have observed that the results are not the same in all patients. The reason may lie in the gut bacteria.
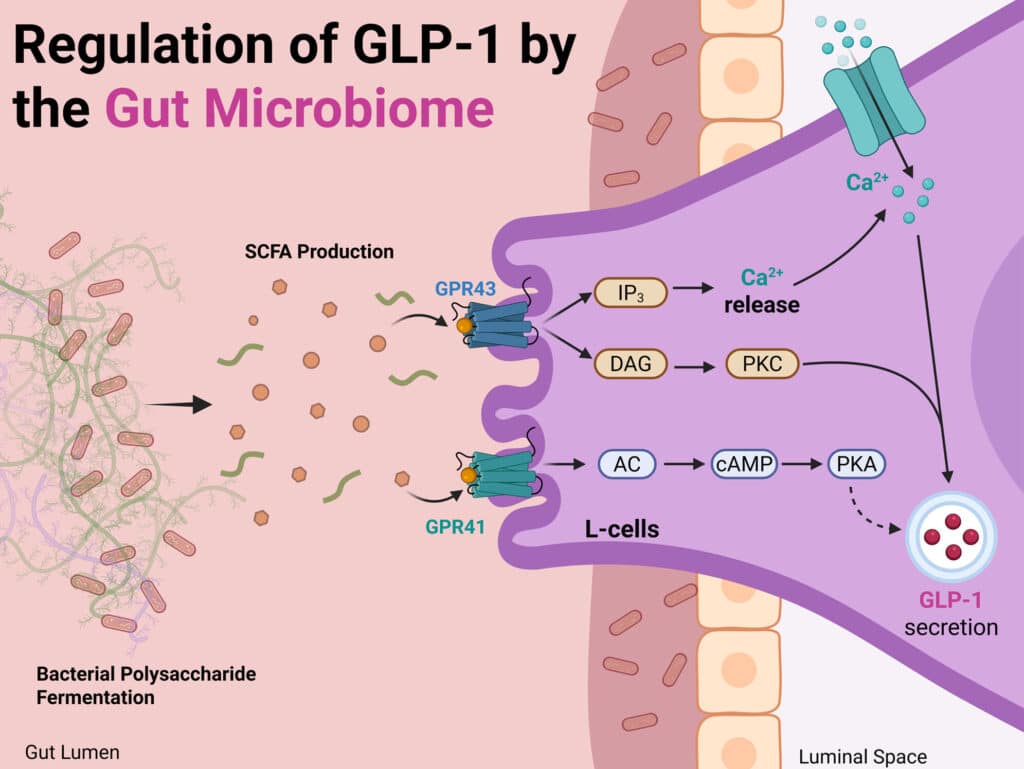
How bacteria can influence metabolism
When we consume foods rich in fibre, certain intestinal bacteria transform them into molecules that stimulate the natural release of GLP-1..
In other words: the microbiome can enhance or weaken the metabolic signal on which these drugs act.
In addition, when there is a microbial imbalance - a relatively common occurrence in modern life - low-grade inflammation appears, a phenomenon that researchers link to:
- insulin resistance
- worse metabolic response
- increased risk of age-related diseases.
That is why some scientists believe that the gut could be directly influencing the efficacy of treatments that are now considered key to longevity medicine.
What is interesting is that these two stories - a bacterium discovered decades ago and the most advanced drugs of the day - reflect the same change in the way we understand the human body. Medicine is shifting from treating isolated organs to studying internal biological ecosystems. And in this new map, the gut occupies a central position.
A growing body of research suggests that factors such as diet, stress, digestive infections or long-term use of medications can modify the microbiome and thereby alter metabolic processes that influence long-term health.
Trend-setting scientific insights
- The microbiome may explain individual differences in response to metabolic treatments.
- Chronic low-grade digestive inflammation is being linked to diseases associated with ageing.
- Modern metabolic drugs do not only act on the body: they also appear to modify the gut ecosystem.
- In the future, medicine could combine drug treatments with strategies targeting the microbiome.
The research is still developing, but the message emerging is increasingly clear: the digestive system not only influences how we digest food, but also how we respond to diseases and treatments.
From silent infections such as Helicobacter pylori to innovative therapies for obesity, science is revealing that the gut may be one of the great regulators of human health. And probably also longevity.